 Sugars: The Difference Between Fructose, Glucose And Sucrose CH2OHCH(OH)CHO is oxidised by Benedict's reagent to CH2OHCH(OH)COOH the aldehyde group is oxidised to It is a reducing sugar and gives a positive test with Benedict's reagent. The presence of an aldehyde group means that glyceraldehyde can also be classified as an aldose. Its structural formula shows it contains an aldehyde group (-CHO) and two hydroxyl groups (-OH). Glyceraldehyde's molecular formula is C3H6O3. Molecules that have the same molecular formula but different structural formulae are called structural isomers. fructose, glucose and galactose ('hex' indicates 6) There is more than one molecule with the molecular formula C5H10O5 and more than one with the molecular formula C6H12O6. ribose and deoxyribose ('pent' indicates 5) n = 6 hexoses, e.g. They can be classified according to the number of carbon atoms in a molecule: n = 3 trioses, e.g. Monosaccharides have the general molecular formula (CH2O)n, where n can be 3, 5 or 6. They are the building blocks from which all bigger carbohydrates are made. cellulose in plants and glycogen in the human body components of other molecules eg DNA, RNA, glycolipids, glycoproteins, ATP Monosaccharides Monosaccharides are the simplest carbohydrates and are often called single sugars. starch in plants building blocks for polysaccharides (giant carbohydrates), e.g. Carbohydrates are: a source of energy for the body e.g. Other carbohydrate molecules are very large (polysaccharides such as starch and cellulose). Sucrase, lactase, and maltase are located on the outer surfaces of epithĬarbohydrates (also called saccharides) are molecular compounds made from just three elements: carbon, hydrogen and oxygen. Maltose comes from the hydrolysis of starch and is in turn hydrolyzed to glucose by maltase. In maltose, two glucose units are joined by an α-1,4 glycosidic linkage, as stated earlier. Lactose is hydrolyzed to these monosaccharides by lactase in human beings (Section 16.1.12) and by β-galactosidase in bacteria. Lactose, the disaccharide of milk, consists of galactose joined to glucose by a β-1,4-glycosidic linkage. Sucrose can be cleaved into its component monosaccharides by the enzyme sucrase. The anomeric carbon atoms of a glucose unit and a fructose unit are joined in this disaccharide the configuration of this glycosidic linkage is α for glucose and β for fructose. Sucrose (common table sugar) is obtained commercially from cane or beet. Three abundant disaccharides are sucrose, lactose, and maltose (Figure 11.11). Sucrose, Lactose, and Maltose Are the Common Disaccharides A disaccharide consists of two sugars joined by an O-glycosidic bond. Indeed, the wide array of these linkages in concert with the wide variety of monosaccharides and their many isomeric forms makes complex carbohydrates information-rich molecules. 
The fact that monosaccharides have multiple hydroxyl groups means that various glycosidic linkages are possible. Such a linkage is called an α-1,4-glycosidic bond. 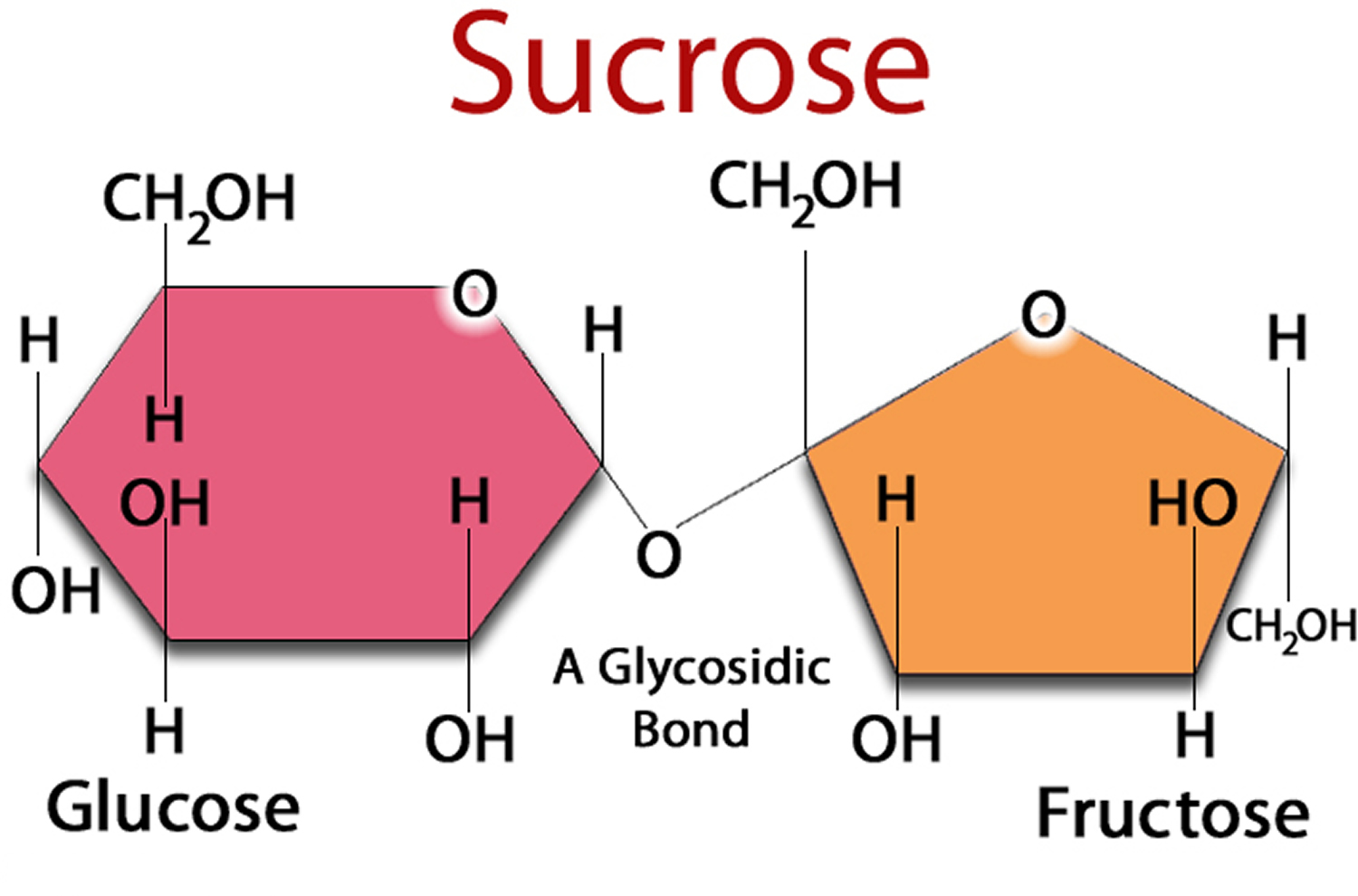 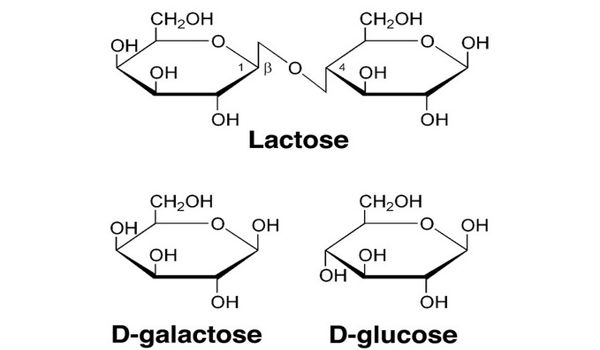
In maltose, for example, two d-glucose residues are joined by a glycosidic linkage between the α-anomeric form of C-1 on one sugar and the hydroxyl oxygen atom on C-4 of the adjacent sugar. Oligosaccharides are built by the linkage of two or more monosaccharides by O-glycosidic bonds (Figure 11.10). Complex Carbohydrates Are Formed By Linkage Of Monosaccharidesīecause sugars contain many hydroxyl groups, glycosidic bonds can join one monosaccharide to another. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
